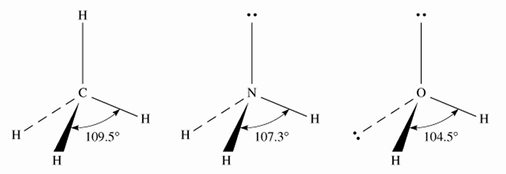
10.1 Molecular Geometry
Give the number of lone pairs around the central atom and the molecular geometry of NH4+.
1) 0 lone pairs, tetrahedral
2) 1 lone pair, trigonal bipyramidal
3) 1 lone pair, seesaw
4) 2 lone pairs, T-shaped
5) 3 lone pairs, trigonal bipyramidal
Give the number of lone pairs around the central atom and the molecular geometry of the tri-iodide ion, I3-.
1) 0 lone pairs, trigonal planar
2) 1 lone pair, tetrahedral
3) 1 lone pair, trigonal pyramidal
4) 2 lone pairs, seesaw
5) 3 lone pairs, linear
According to VSEPR theory, which of the following species should have a tetrahedral molecular geometry?
1) H2O
2) NH3
3) AlF3
4) CCl4
5) CH2O
Which pair of molecules has the same molecular geometry?
1) CCl4 and NH3
2) H2O and SO2
3) NO3 and H2O
4) CO2 and CCl4
5) SO2 and NH3
According to the VSEPR theory, if the central atom has three bonded atoms and one lone pair,
the geometry (shape) at this atom will be ______________________.
1) linear
2) bent (angular)
3) trigonal planar
4) trigonal pyramidal
5) tetrahedral
Which of the following substance is/are planar?
(i) SO3 (ii) SO32- (iii) NO3- (iv) PF3 (v) BF3
1) only (i) and (ii)
2) only (i), (iii), and (v)
3) only (iv)
4) all are planar except (iv)
5) all are planar except (ii)
According to the VSEPR theory, the actual F-As-F bond angles in the AsF4- ion are predicted to be
1) 109.5°.
2) 90° and 120°.
3) 180°.
4) < 109.5°.
5) < 90° and < 120°.
Predict the geometry around the central atom in SO42-.
1) trigonal planar
2) trigonal pyramidal
3) tetrahedral
4) trigonal bipyramidal
5) octahedral
List the following molecules in order of increasing bond angle (smallest to largest):
NH3, BF3, ClNO, CF4
1) NH3, BF3, ClNO, CF4
2) ClNO, BF3, NH3, CF4
3) ClNO, CF4, NH3, BF3
4) CF4, NH3, BF3, ClNO
5) NH3, CF4, ClNO, BF3
Give the number of lone pairs around the central atom and the molecular geometry of SCl2.
1) 0 lone pairs, linear
2) 1 lone pair, bent
3) 2 lone pairs, bent
4) 3 lone pairs, bent
5) 3 lone pairs, linear
Give the number of lone pairs around the central atom and the geometry of the ion NO2–.
1) 0 lone pairs, linear
2) 1 lone pair, bent
3) 2 lone pair, bent
4) 3 lone pairs, bent
5) 3 lone pairs, linear
According to the VSEPR theory, which one of the following species should be linear?
1) H2S
2) HCN
3) BF3
4) H2CO
5) SO2
The C–N–O bond angle in nitromethane, CH3
NO2, is expected to be approximately
1) 60°
2) 90°
3) 109.5°
4) 120°
5) 180°
Which of the following molecules have the same geometries?
1) SF4 and CH4
2) CO2 and H2O
3) CO2 and BeH2
4) N2O and NO2 (NNO and ONO, respectively)
According to the VSEPR theory, the molecular shape of the carbonate ion, CO32-, is
1) square planar.
2) tetrahedral.
3) pyramidal.
4) trigonal planar.
5) octahedral.
According to VSEPR theory, which one of the following molecules should have a bent shape?
1. Cl2O
2. CO2
3. HCN
4. CCl4
5. none of them
10.2 Dipole Moments
Which one of the following molecules is nonpolar?
1) NH3
2) OF2
3) CH3Cl
4) H2O
5) BeCl2
Which of the following species has the largest dipole moment (i.e., is the most polar)?
1) CH4
2) CH3Br
3) CH2Br2
4) CHBr3
5) CBr4
10.4 Hybridization of Atomic Orbitals
Indicate the type of hybrid orbitals used by the C atom indicated by the arrow in the molecule below.
1) sp
2) sp2
3) sp3
4) sp3d
5) sp3d2
In which one of the following molecules is the central atom sp2 hybridized?
1. SO2
2. N2O
3. BeCl2
4. NF3
5. PF5
What is the hybridization of the central atom in ClO3-?
1. sp
2. sp2
3. sp3
4. sp3d
5. sp3d2
10.5 Hybridization in Molecules Containing Double and Triple Bonds
The number of pi bonds in this molecule is:
1) 2
2) 3
3) 4
4) 5
5) 6
The skeleton for methyl formate (C2H4O2) is given below. Complete the following statement: Methyl formate has ______ pi bond(s); carbon #1 has _____ hybridization, while carbon #2 has _______ hybridization.

1) 1, sp2, sp3
2) 1, sp3, sp2
3) 3, sp2, sp3
4) 2, sp, sp3
5) 2, sp2, sp3
The number of pi bonds in the molecule below is
1) 2
2) 4
3) 6
4) 10
5) 15
Indicate the type of hybrid orbitals used by the central atom in CCl4.
1) sp
2) sp2
3) sp3
4) sp3d
5) sp3d2
The number of pi bonds in the molecule below is
1. 1.
2. 2.
3. 3.
4. 5.
5. 9.
Key:
10.1: 1, 5, 4, 2, 4, 2, 5, 3, 5, 3, 2, 2, 4, 3, 4, 1
10.2: 5, 4
10.3:
10.4: 1, 1, 3
10.5: 5, 1, 2, 3, 3